thermodynamics - Confused why delta ∆Q and dQ don't make sense for heat Q - Chemistry Stack Exchange
Por un escritor de hombre misterioso
Descripción
In my chemistry teacher's notes, some notations concerning the heat $Q$ are marked as inappropriate.
$Q$: yes
d$Q$: no
$\delta Q$: yes
$\Delta Q$: no
In the second bullet in the screenshot below

Tackling a Century Mystery: Entropy
What is the basic difference between the heat of formation and the heat of reaction? - Quora
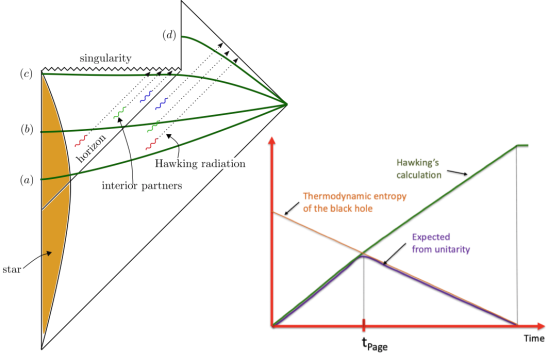
Ro's blog Musings of a nascent physicist

Falkovich Statistical - Physics Notes, PDF, Heat

PhD Thesis Zino Boisdenghien

Can the Second law of thermodynamics be abandoned?

First Law of Thermodynamics

Full article: Theory of cross phenomena and their coefficients beyond Onsager theorem

First Law of Thermodynamics

105 questions with answers in STATISTICAL PHYSICS

General Chemistry/Print version - Wikibooks, open books for an open world

Clausius Inequality says the integral of dq/T for a process is less than equal to zero. Also any process needs to have ∆s=dq/T >0. Isn't this contradictory? - Quora

Falkovich Statistical - Physics Notes, PDF, Heat
Why is 'enthalpy change' (∆H) not always equal to 'heat transfer' (Q)? - Quora

Tackling a Century Mystery: Entropy
de
por adulto (el precio varía según el tamaño del grupo)







